 Download: Click Here
Download: Click Here
Below are pictures for preview:
Practical Guides for CAIE IGCSE and A Level Exams


今天是A2 Biology的前两个章节,呼吸作用和光合作用。它们都是能量的转化过程,有很多可以做对比的地方。因此,我把它俩放到一块儿了。PDF下载链接请戳这里。
以下是预览:
Today, we present ‘Chemistry of Organic Compounds’, which collects reactions of organic compounds included in the CAIE 9701 A Level Chemistry syllabus. It does not include aromatic compounds, however, which has been published as a separate issue.
To download the PDF version, please click here.
Below are pictures for preview.
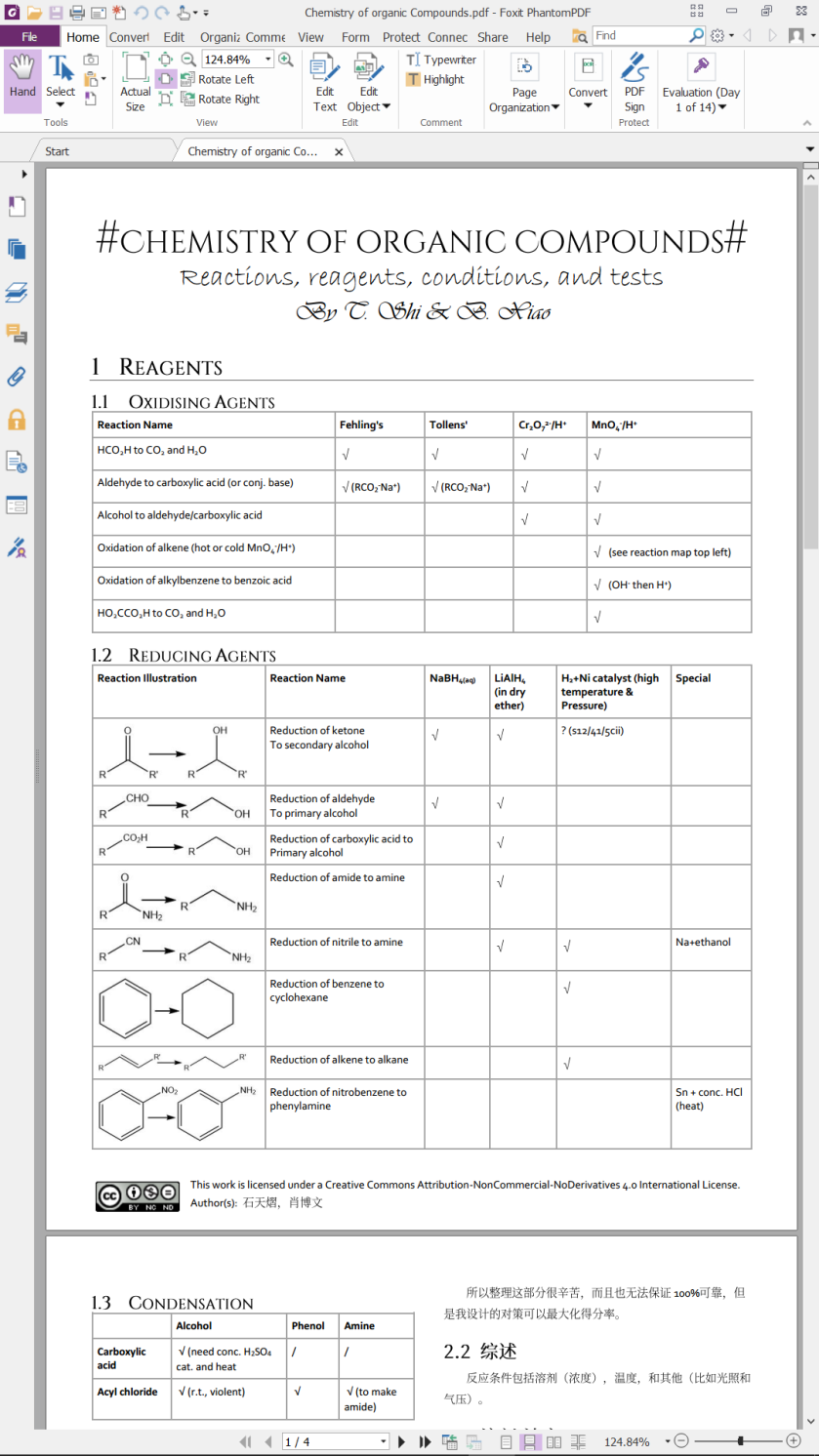

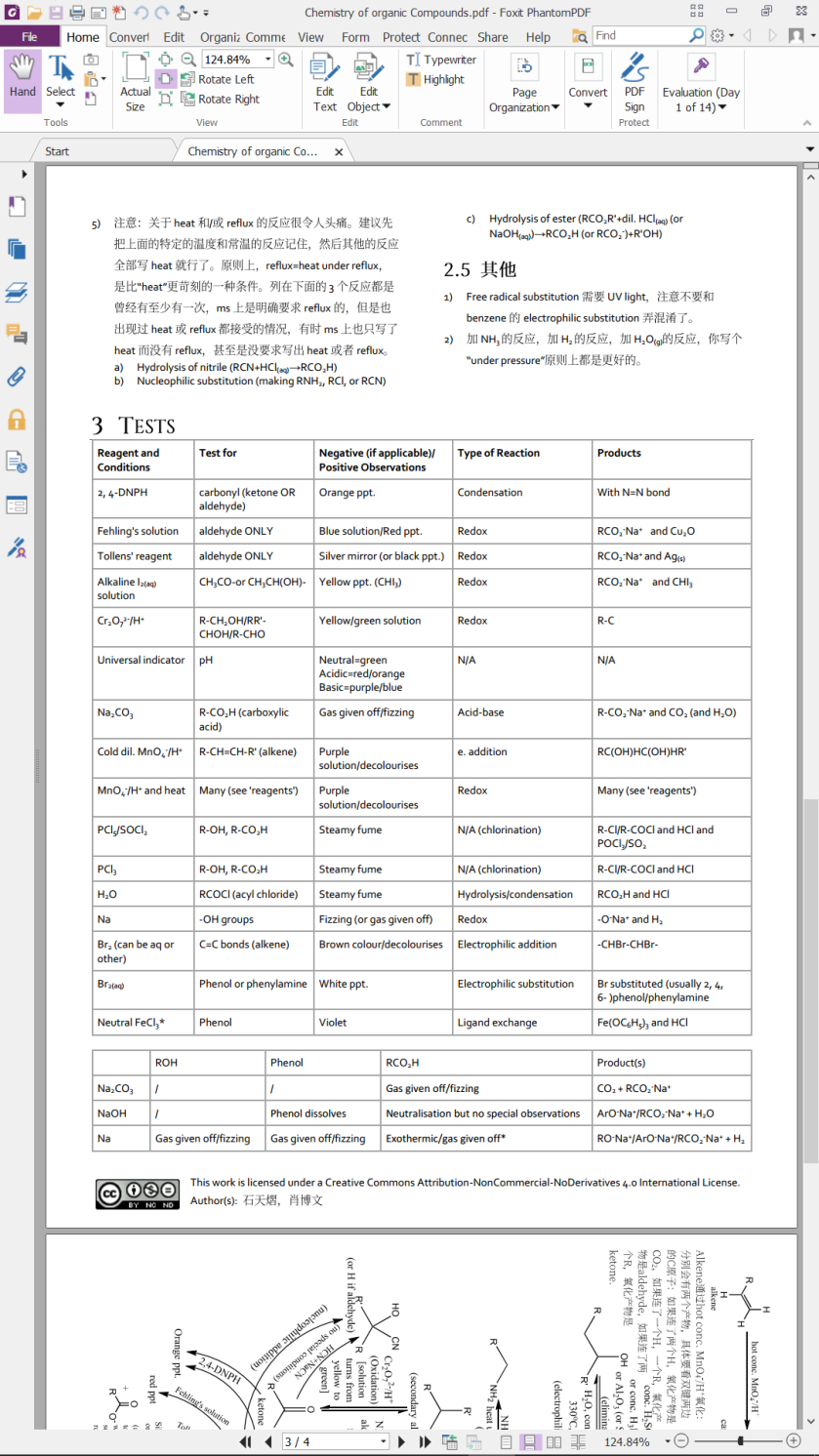


This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
本文作者/Authour(s):肖博文,石天熠
CAIE IAL化学有一类题目是结构式(structural formula)和分子式(molecular formula)的转换。
从结构式(一般是skeletal formula)到分子式的转换,虽然是考察最基础的知识,但是经常会因为粗心大意而写错。最常见的原因就是数漏H原子,尤其是遇到较大的,带环的,有多个带H的基团的分子的时候。
从分子式到结构式的转换,属于分析化学的范畴。给你一个分子式,然后告诉你这个分子和特定的几个试剂的反应情况,或是给你图谱分析。
不饱和度(degree of unsaturation)的使用可以有效地帮助解答这两种题。今天介绍第一种,即从结构式到分子式。
我们知道,hexane的molecular formula是C6H14,H的数量=2*C的数量+2.
你可以试着把它的isomers画出来,比如(CH3)2CHCH2CH2CH3, (CH3)3CCH2CH3, (CH3)2CHCH(CH3)2, 你会发现,hexane和它的isomers,全部都是无环,且全部是单键(即饱和)的结构。
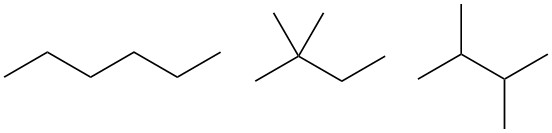
那么如果出现了双键和环呢?
还是6个C,这次我们试着加双键和/或环。
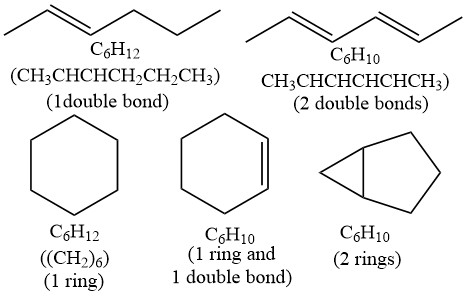
我们发现,每加一个双键或环,H的数量就比原来饱和的状态(即hexane,C6H14)少2个。
我们把双键+环的数量叫做不饱和度(Degree of unsaturation,DU)。
于是在碳氢化合物中,H的数量=2*C的数量+2-2*DU。
有一个特殊情况是苯环。我们熟悉的苯环结构是六边形+圆形(Thiele结构),这使得我们无法直观看出苯环的不饱和度。因此,我们看苯环的Kekulé结构:
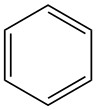
可以看到,苯环等同于一个有3个双键和1个环的6碳的碳氢化合物。
6碳的饱和状态是6*2+2=14个H,而苯环的DU=3+1=4,H减少2*4=8个,H的实际数量是14-8=6个。
的确,苯的molecular formula就是C6H6.
但是显然CAIE不会出这么简单的题。O,N,和/或卤素原子必然会出现在这类题型。
怎么办呢?
我们再找规律。
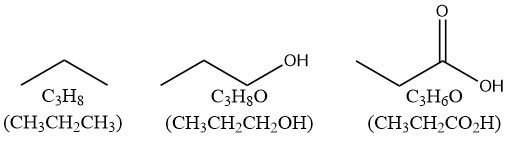
我们知道propane的molecular formula是C3H8,它全部是单键。我们给它加个-OH,变成propanol,molecular formula是C3H8O。H的数量仍然是8,所有的共价键也全部是单键。
那我们在O上做一个双键试试?我们得到了Propanoic acid,它的H数量减少了2个。这说明O是不影响H的数量的,但是O上的双键,和C上的双键一样,都会减少2个H的数量。
类似地,我们来研究N和卤素原子对H数量的影响:
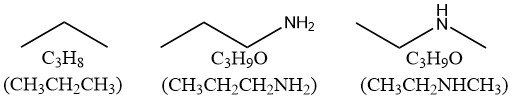
我们发现,1个N原子会使H的数量增加1个。
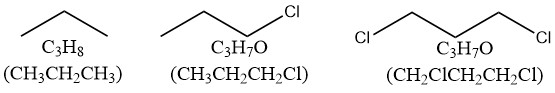
而1个卤素原子会使H的数量减少一个。
于是我们便知道了所有的规律:
饱和状态下,H的数量=C的数量*2+2。
每个双键和环会减少2个H的数量。
O原子不影响H的数量。N原子会增加1个H的数量,卤素原子会减少1个。
根据这些规律,我们可以列出一个从结构式转换分子式的protocol:
第一步,数C的数量。
第二步,计算饱和状态时对应的H的数量,即2*C的数量+2.
第三步,计算不饱和度(DU)和H的减少量。其中不饱和度=双键的数量+环的数量,H的减少量=2*不饱和度。
第四步,寻找N和卤素原子,计算H的变化量。N增加一个H原子,卤素减少一个H原子。
第五步,计算实际H的数量,即第二步算出的数量–第三步算出的数量+第四部算出的变化量。
第六步,写出molecular formula,注意不要漏掉O,N和卤素原子。
第一题
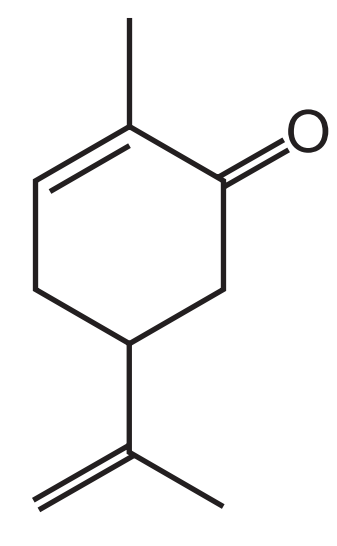
答案:
第一步,数C的数量:六元环6个,加上顶部1个和底部3个,共10个。
第二步,计算饱和状态时对应的H的数量:是2*10+2=22个。
第三步,计算不饱和度(DU)和H的减少量:3个双键,1个环,DU=4,H减少2*4=8个。
第四步,寻找N和卤素原子,计算H的变化量:无N和卤素原子,H的变化量=0.
第五步,计算实际H的数量:22-8+0=14.
第六步,写出molecular formula:C10H14O
第二题
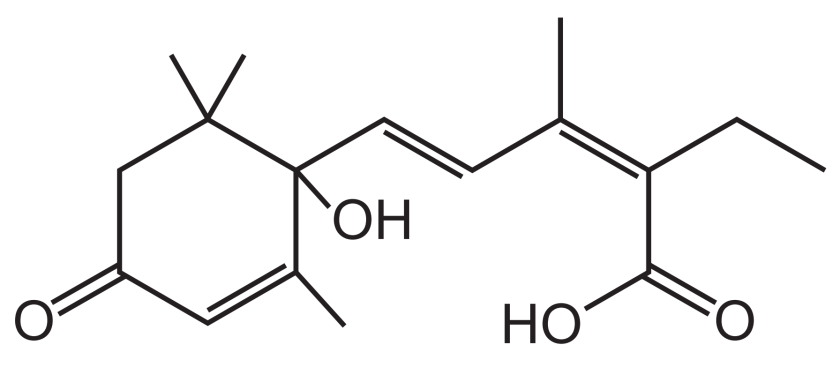
答案:
第一步,数C的数量:6+2+1+(6+1+1)=17个
第二步,计算饱和状态时对应的H的数量:是2*17+2=36个。
第三步,计算不饱和度(DU)和H的减少量:5个双键,1个环,DU=6,H减少2*6=12个。
第四步,寻找N和卤素原子,计算H的变化量:无N和卤素原子,H的变化量=0.
第五步,计算实际H的数量:36-12+0=24.
第六步,写出molecular formula:C10H24O4
第三题
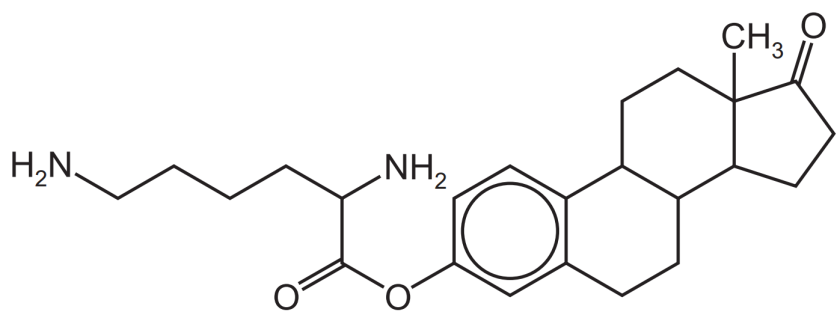
答案:
第一步,数C的数量:(注意环之间的C是有重复的,不要直接6*3+5!)一共24个。
第二步,计算饱和状态时对应的H的数量:是2*24+2=50个。
第三步,计算不饱和度(DU)和H的减少量:5个双键,4个环,DU=9,(或者1个苯环(DU=4),3个环,2个双键,DU=4+3+2=9)H减少2*9=18个。
第四步,寻找N和卤素原子,计算H的变化量:有2个N,无卤素原子,H的变化量=+2.
第五步,计算实际H的数量:50-18+2=34.
第六步,写出molecular formula:C24H34N2O3
注意,严谨地来说,DU是π bonds的数量(双键有一个bond,叁键有两个π bonds)+环(仅桥环;螺环化合物不一样)的数量。因为CAIE目前出现的这类题,仅仅包括了双键和桥环化合物,所以我没深入讨论更多的情况。

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
© 2018 by CAIEhelp,a division of Brilas Academy (萤火);Author of this work: 石天熠


This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
本文作者/Authour(s):石天熠,肖博文



Today, we present ‘Chemistry of Organic Compounds’, which collects reactions of organic compounds included by the CAIE 9701 A Level Chemistry syllabus. It do not include aromatic compounds, however, which has been published as a separate issue.
To download the PDF version, please click here.
Below are pictures for preview.





This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
本文作者/Authour(s):张家栋,石天熠


This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
本文作者/Authour(s):张家栋,石天熠
公式
首先我们来看一下公式。因为LaTeX格式与Word内置的公式格式不兼容,所以用图片的形式发出来。
1.radian: the angle subtended at the centre of a circle(1) by an arc of length equal to radius of the circle(1).
2.angular velocity: the angle swept out by the radius per second(1).
3.centripetal force:the net force acting on an object moving in a circle(1), it is always directed towards the centre of circle(1).
4.gravitational field: region of space where mass experience a force(1).
5.point mass:the size of the object is extremely small comparing with their separation.
6.Newton’s law of gravitation: two point masses attract each other with a force that is directly proportional to the product of their masses(1) and inversely proportional to the square of their separation(1).
7.gravitational field strength:the force per unit mass
8.gravitational potential:the work done in bringing unit mass from infinity to that point.
1.Similarity and difference between electric and gravitational field.
Similarity: radial field, force inversely proportional to square of separation(1)
Differences: gravitational field always attractive, electric field could be attractive or repulsive(1).
2. What is meant by field of force: region in space where a particle experiences a force.
3. Why is gravitational potential negative?
Potential at infinity is defined as zero(1), gravitational force is attractive so work is done by the object as object moves(1).
4. A calculation problem associated with a graph, what is the min speed to reach another star?
In this situation, the change in potential should be potential difference to the highest point, rather than the potential difference between start and final point.
5. Start from the moon, why the min speed to reach earth is different from the min speed to reach infinity?
At earth, the potential due to the moon is not zero, it is also attracted by the earth(1), so the min escape speed would be lower(1).
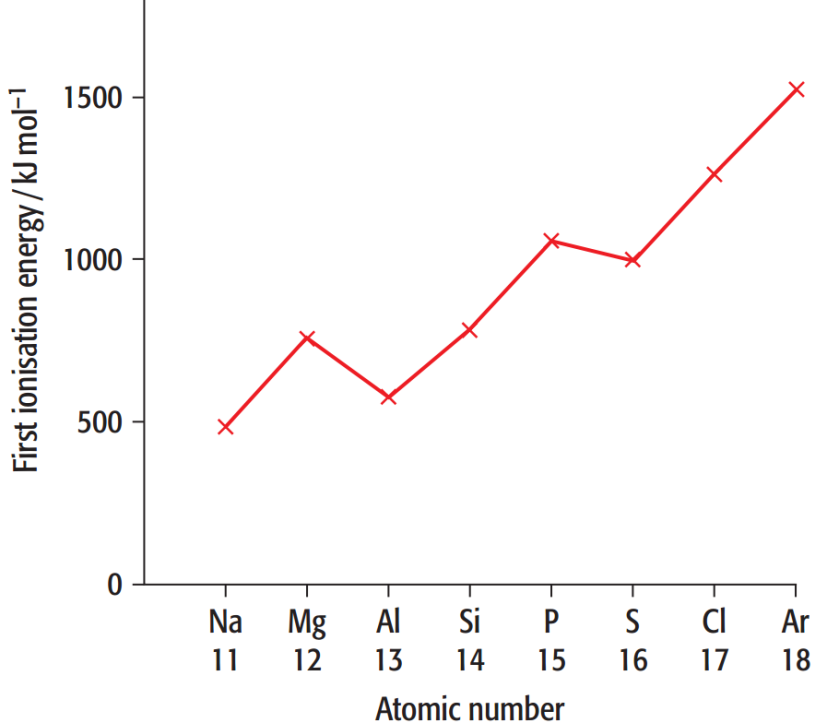 Sodium and Magnesium
Sodium and Magnesium





This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
本文作者:石天熠

All contents are concordant with mark schemes of CAIE 9700 Int’ A Level Biology past paper 4s from year 2014 to 2017 unless being noted with an asterisk (*).

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
本文作者/Authour(s):石天熠,肖博文
Homeostasis in Humans in General
A process in which a change in some parameter, such as blood glucose level, brings about processes which move its level back towards normal again.
In response, hormones are released and they reach their target organs, or effectors.
Homeostasis is the maintenance of constant internal environment irrespective of changes in external environment, using negative feedback mechanisms. This involves the initial stimulus, which is the change in a parameter detected by receptors, and the action taken by the effectors, allowing restoration of the ‘set point’. There is fluctuation around the norm. Examples of homeostasis include thermoregulation, osmoregulation, and blood glucose regulation.
Homeostasis is the maintenance of constant internal environment, for example several parameters of blood (e.g. blood glucose level, osmolarity and blood pressure), causing them to fluctuate within narrow ranges around the set point.
Homeostasis of body temperature, water potential (osmolarity) and blood glucose level is critical. Low body temperature leads to slowed metabolism and enzyme activity, while high temperature causes enzymes to be denatured; low water potential causes cells to shrink while high water potential causes cells to burst; low blood glucose level reduces cellular respiration while high blood glucose level leads to lowered blood water potential, which further leads to shrinkage of cells.
Human Kidney Anatomy and Physiology
Tendency of water molecules to move from one region to another
The diameter of afferent arterioles is greater than that of the efferent arterioles, so this builds up a high blood pressure in the glomerulus and molecules tend to move from the capillary into the Bowman’s capsule.
Molecules first pass through holes in the endothelium, then the basement membrane, and finally gaps between podocytes and enter Bowman’s capsule. The basement membrane is selectively permeable: only molecules with molecular weight less than 69,000 can pass through it.
1. Proteins are hydrolysed into amino acids in gut
2. Excess amino acids cannot be stored
3. Amino acids are deaminated in liver to produce urea
4. Urea in blood is filtered into nephrons by ultrafiltration
The hypothalamus has neurosecretory cells, which produce ADH, and osmoreceptors, which detects changes in water potential of the blood.
Osmoreceptors shrink when water potential is low, and this causes ADH to be released from the posterior pituitary.
Ammonium ions/ammonia is produced in the liver from excess amino acids, which is toxic to the body if they are allowed to accumulate. Ammonium ions/ammonia combine with carbon dioxide to form urea in urea (ornithine) cycle.
Hormonal Control of Female Reproductive System
1. It is mostly secreted during the second half of the cycle (i.e. from day 14 onwards)
2. It maintains the thickness of the endometrium in preparation for implantation
4. inhibits GnRH production and hence development of new follicle
1. It stimulates follicle to secrete oestrogen
3. Surge in LH secretion stimulates ovulation
5. It stimulates development of corpus luteum, which secretes progesterone
1. Thickening of endometrium
2. Development of capillaries in the endometrium
3. Inhibition of FSH production
 Blood Glucose Level
Blood Glucose Level
Homeostasis in Plants
Plants secrete abscisic acid at times of water stress. It closes the stomata to reduce water loss.
Abscisic acid binds to receptors on cell surface membrane of guard cells, inhibiting proton pumps. H+ concentration inside the cell increases.
Abscisic acid also stimulates the influx of Ca2+ ions, which act as a second messenger to activate channel protein to open, encouraging efflux of K+ ions.
Water potential of the cells increases, so water leaves cells by osmosis. Volume of the guard cells decrease and cells become flaccid.
Thermoregulation